teaser
Kristine Breuer
MD
Thomas Werfel
MD
Alexander Kapp
MD PhD
Department of Dermatology and Allergology
Hannover Medical University
Germany
E:[email protected]
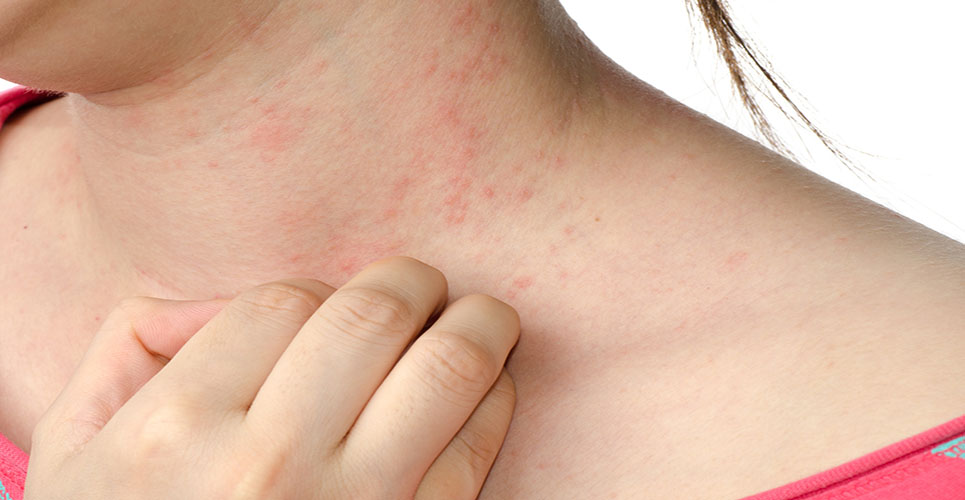
Atopic dermatitis (AD) is a chronic inflammatory skin disease that often begins in early infancy and runs a course of remission and exacerbation. Acute eczematous lesions are characterised by erythema, oozing and crusting, whereas chronic lesions show thickened skin and papules. Dry skin, pruritus and sleeplessness are other characteristics of AD.(1) Various factors triggering AD have been identified, such as food allergens, inhalative allergens, bacterial superantigens and psychological stress.(2–5) Approximately 10–20% of infants in Western countries suffer from AD, and the prevalence seems to be increasing,(6,7) with 85% of affected children presenting the first lesions by the age of five years.(8)
In patch test and spontaneous lesions, the cellular infiltrate is dominated by CD4-positive T-helper cells. Mononuclear cells and eosinophil granulocytes can mainly be found in the dermis,(9,10) Langerhans cells (LCs) with specific IgE bound to Fce-RI on their surface in the epidermis.(11,12) Whereas the Th2-like cytokine IL-4 plays a role in the initial phase of cutaneous inflammation, the Th1-like cytokine IFN-gamma predominates in spontaneous lesions or older patch test lesions in AD.(13–15) Topical corticosteroids have been the treatment of choice for flares of AD for decades, but potential side-effects have limited their use. As no widely accepted treatment for flares of AD was available, the development of new topical agents for the treatment of cutaneous inflammatory lesions was required, particularly for long-term treatment.
Immunosuppressive macrolides
Various strains of Streptomyces produce immunosuppressive macrolides, small molecules that are able to penetrate lesional skin, thereby exhibiting effects on the cells of the cutaneous immune system without inducing systemic side-effects. Immunosuppressive macrolides were initially developed to prevent the rejection of transplanted organs, before their benefit in the treatment of inflammatory dermatological conditions was discovered.
Cyclosporine, which is an effective drug for the systemic treatment of severe AD, does not penetrate the epidermis when used as a topical formulation. Due to potential toxic side-effects, such as nephrotoxicity and hypertension, that may occur upon systemic application, cyclosporine is used only in severe cases of AD.
Tacrolimus, also known as FK506, was isolated from a culture of Streptomyces tsukubaensis.(16) It was the first macrolide shown to penetrate the epidermis of inflamed skin in significant amounts (due to a relatively small molecular weight of 822 Da) and to be effective in the treatment of AD. In contrast, tacrolimus cannot penetrate normal skin. The ascomycin derivative pimecrolimus, also known as SDZ ASM 981, has a similar structure and a molecular weight of 810 Da.(17)
T-cells are activated via calcium-dependent pathways in which calcineurin dephosphorylates the nuclear factor of activated T-cells (NF-AT). NF-AT translocates to the nucleus, which leads to the transcription of various cytokines.(18)
Tacrolimus and pimecrolimus interact with macrophilin-12 (FKBP12), a cyclophilin-like cytoplasmic protein, and the resulting complex inhibits the ability of calcineurin to dephosphorylate the transcription factor NF-AT.(19–21) Since only dephosphorylated NF-AT can translocate into the nucleus, the transcription of various proinflammatory cytokines and other mediators of the allergic inflammatory reaction is inhibited.(18)
Although the mechanism of action of tacrolimus is similar to that of cyclosporine, which binds to the immunophilin cyclophilin, the potency of tacrolimus to inhibit T-cell activation is 10–100 times greater.(16)
Pimecrolimus and tacrolimus are able to inhibit the early activation and proliferation of human T-cells by downregulation of IL-2 at nanomolar concentrations. Furthermore, the production of Th1- and Th2-type cytokines is downregulated.(20)
In addition to their impact on T-cells, immunomodulatory macrolides have an influence on mast cells. Both SDZ ASM and tacrolimus inhibit the FceRI-mediated activation of human mast cells in vitro, through a mechanism that involves binding to FKBP12.(22–24) These effects might explain the fast relief of pruritus and erythema that occurs shortly after initiation of the treatment.
Studies on human antigen-presenting cells have been performed with tacrolimus only, and an influence on epidermal dendritic cells has been demonstrated. In an in-vivo intervention study, the topical application of tacrolimus led to a decrease of CD1a+ inflammatory dendritic epidermal cells (IDECs) in the epidermal dendritic cell population. Moreover, expression of FceRI and CD36 was downregulated on both IDECs and LCs,(25) an effect that might explain the stronger anti-inflammatory effect shown in clinical studies, compared with pimecrolimus. In addition, the expression of IL-2R and of costimulatory molecules CD80 and CD40 was found to be downregulated on LCs in vitro, thus leading to a decrease of the stimulatory capacity of LCs.(26)
Although tacrolimus has no direct antistaphylococcal activity,(27) an overall decrease of colonisation density was observed by the first week of treatment with 0.1% tacrolimus ointment in patients with AD, while the eczema score improved in parallel (which may be a consequence of the improved skin barrier). Moreover, tacrolimus blocked S aureus superantigen-induced T-cell proliferation in the peripheral blood mononuclear cells of patients with AD and in healthy individuals (control group).(28) The paradoxical eradication of S aureus from the skin, which was also observed following treatment with topical corticosteroids, might therefore be due to the healing of inflammatory lesions.
Side-effects, including signs of irritation such as erythema and burning, may occur upon use of topical immunomodulators in patients with AD. No significant increase of skin or systemic infections was observed in clinical studies performed in children and adults with AD, and no decrease in recall antigen reactions was induced. However, in a photocarcinogenesis study in hairless mice treated with tacrolimus, the median time to tumour onset was decreased; however, these observations may not be transferable to humans.
Other cutaneous diseases, such as pyoderma gangrenosum,(29) contact dermatitis,(30) dyshidrotic eczema,(31) lupus erythematosus,(32) psoriasis (under occlusion),(33) lichen planus,(34) vitiligo,(35) cutaneous sarcoidosis(36) and graft-versus-host disease(37) also respond to treatment with macrolides, although the studies carried out are either noncontrolled studies or case reports. No placebo-controlled trials have yet been carried out. In addition, recent reports suggest the efficacy of topical immunomodulators in adult patients with seborrheic dermatitis.(38,39)
Conclusion
Taken together, the topical immunomodulators have an impact on multiple cell types and targets that contribute to inflammation in the skin of patients with AD. These effects may explain the profound clinical efficacy of pimecrolimus and tacrolimus observed in the various clinical studies performed in infants, children and adults with AD.
References
- Kapp A. Atopic dermatitis – the skin manifestation of atopy. Clin Exp Allergy 1995;25:210-9.
- Breuer K, Häussler S, Kapp A, et al. Staphylococcus aureus: colonizing features and influence of an antibacterial treatment in adults with atopic dermatitis. Br J Dermatol 2002;147:55-61.
- Breuer K, Heratizadeh A, Wulf A, et al. Late eczematous reactions to food in children with atopic dermatitis. Manuscript submitted.
- Breuer K, Wulf A, Constien A, et al. Birch pollen related food as a provocation factor of atopic dermatitis in childhood. Manuscript submitted.
- Werfel T, Kapp A. Environmental and other major provocation factors in atopic dermatitis. Allergy 1998;53:731-9.
- Schultz Larsen F, Diepgen T, Svensson A. The occurrence of atopic dermatitis in north Europe: an international questionnaire study. J Am Acad Dermatol 1996;34:760-4.
- Williams HC. Is the prevalence of atopic dermatitis increasing? Clin Exp Dermatol 1992;17:385-91.
- Rajka G. Essential aspects of atopic dermatitis. Berlin: Springer Verlag; 1989.
- Bruijnzeel PL, Kuijper PH, Kapp A, et al. The involvement of eosinophils in the patch test reaction to aeroallergens in atopic dermatitis: its relevance for the pathogenesis of atopic dermatitis. Clin Exp Allergy 1993;23:97-109.
- Tanaka Y, Anan S, Yoshida H. Immunohistochemical studies in mite antigen-induced patch test site in atopic dermatitis. J Dermatol Sci 1990;1:361-8.
- Bieber T, de la Salle H, Wollenberg A, et al. Human epidermal Langerhans cells express the high affinity receptor for immunoglobulin E (FceRI). J Exp Med 1992;175:1285-90.
- Mudde GC, von Reijsen FC, Boland GJ, et al. Allergen presentation by epidermal Langerhans’ cells from patients with atopic dermatitis is mediated by IgE. Immunology 1990;69:335-41.
- Grewe M, Gyufko K, Schopf E, et al. Lesional expression of interferon-gamma in atopic eczema. Lancet 1994;343:25-6.
- Werfel T, Morita A, Grewe M, et al. Allergen specificity of skin-infiltrating T-cells is not restricted to a type-2 cytokine pattern in chronic skin lesions of atopic dermatitis. J Invest Dermatol 1996;107:871-6.
- Thepen T, Langeveld-Wildschut EG, Bihari IC, et al. Biphasic response against aeroallergen in atopic dermatitis showing a switch from initial TH2 response into a TH1 response in situ. J Allergy Clin Immunol 1996;97:828-37.
- Goto T, Kino T, Hatanaka H. FK506: historical perspectives. Transplant Proc 1991;23:2713-7.
- Paul C, Graeber M, Stuetz A. Ascomycins:promising agents for the treatment of inflammatory skin diseases. Expert Opin Invest Drugs 2000;9:69-77.
- Kiani A, Rao A, Aramburu J. Manipulating immune responses with immunosuppressive agents that target NFAT. Immunity 2000;12:359-72.
- Grassberger M, Baumruker T, Enz A, et al. A novel anti-inflammatory drug, SDZ ASM 981, for the treatment of skin diseases: in vitro pharmacology. Br J Dermatol 1999;141:264-73.
- Liu J, Farmer JD, Lane WS, et al. Calcineurin is a common target of cyclophiline–cyclosporin A and FKBP–FK506 complexes. Cell 1991;66:807-15.
- Schreiber SL, Crabtree GR. The mechanism of action of cyclosporine A and FK506. Immunol Today 1992;13:136-42.
- de Paulis A, Cirillo R, Ciccarelli A, et al. Characterization of the anti-inflammatory effect of FK506 on human mast cells. J Immunol 1991;147:4278-85.
- de Paulis A, Stellato C, Cirillo R, et al. Anti-inflammatory effect of FK-506 on human skin mast cells. J Invest Dermatol 1992;99:723-8.
- Zuberbier T, Chong SU, Grunow K, et al. The ascomycin macrolactam pimecrolimus (Elidel, SDZ ASM 981) is a potent inhibitor of mediator release from human dermal mast cells and peripheral blood basophils. J Allergy Clin Immunol 2001;108:275-80.
- Wollenberg A, Sharma S, von Bubnoff D, et al. Topical tacrolimus (FK506) leads to profound phenotypic and functional alterations of epidermal antigen-presenting dendritic cells in atopic dermatitis. J Allergy Clin Immunol 2001;107:519-25.
- Panhans-Gross A, Novak N, Kraft S, et al. Human epidermal Langerhans cells are targets for the immunosuppressive macrolide tacrolimus (FK506). J Allergy Clin Immunol 2001;107:345-52.
- Kino T, Hatanaka H, Hashimoto M. FK506 a novel immunosuppressant isolated from a Streptomyces. I. Fermentation, isolation, and physico-chemical and biological characteristics. J Antibiot (Tokyo) 1987;40:1249-55.
- Hauk PJ, Leung DY. Tacrolimus (FK506): new treatment approach in superantigen-associated diseases like atopic dermatitis? J Allergy Clin Immunol 2001;107:391-2.
- Petering H, Kiehl P, Breuer K, et al. Pyoderma gangraenosum: successful topical therapy with tacrolimus (FK506). Hautarzt 2001;52:47-50.
- Queille-Roussel C, Graeber M, Thurston M, et al. SDZ ASM 981 is the first non-steroid that suppresses established nickel contact dermatitis elicited by allergen challenge. Contact Dermatitis 2000;42:349-50.
- Schnopp C, Remling R, Möhrenschlager M, et al. Topical tacrolimus (FK506) and momethasone furoate in treatment of dyshidrotic palmar eczema: a randomized, observer-blinded trial. J Am Acad Dermatol 2002;46:73-7.
- Yoshimasu T, Ohtani T, Sakamoto T, et al. Topical FK506 (tacrolimus) therapy for facial erythematous lesions of cutaneous lupus erythematosus. Eur J Dermatol 2002;12:50-2.
- Mrowietz U, Graeber M, Bräutigam M, et al. The novel ascomycin derivative SDZ ASM 981 is effective for psoriasis when used topically under occlusion. Br J Dermatol 1998;139:992-6.
- Kaliakatsou F, Hodgson TA, Lewsey JD, et al. Management of recalcitrant ulcerative oral lichen planus with topical tacrolimus. J Am Acad Dermatol 2002;46:35-41.
- Grimes PE, Soriano T, Dytoc MT. Topical tacrolimus for repigmentation of vitiligo. J Am Acad Dermatol 2002;47:789-91.
- Katoh N, Mihara H, Yasuno H. Cutaneous sarcoidosis successfully treated with topical tacrolimus. Br J Dermatol 2002;147:154-6.
- Choi CJ, Nghiem P. Tacrolimus ointment in the treatment of chronic cutaneous graft-vs-host disease. Arch Dermatol 2001;137:1202-6.
- Brownell I, Quan LT, Hsu S. Topical pimecrolimus in the treatment of seborrheic dermatitis. Dermatol Online J 2003;9:13.
- Meshkinpour A, Sun J, Weinstein G. An open pilot study using tacrolimus ointment in the treatment of seborrheic dermatitis. J Am Acad Dermatol 2003;49:145-7.