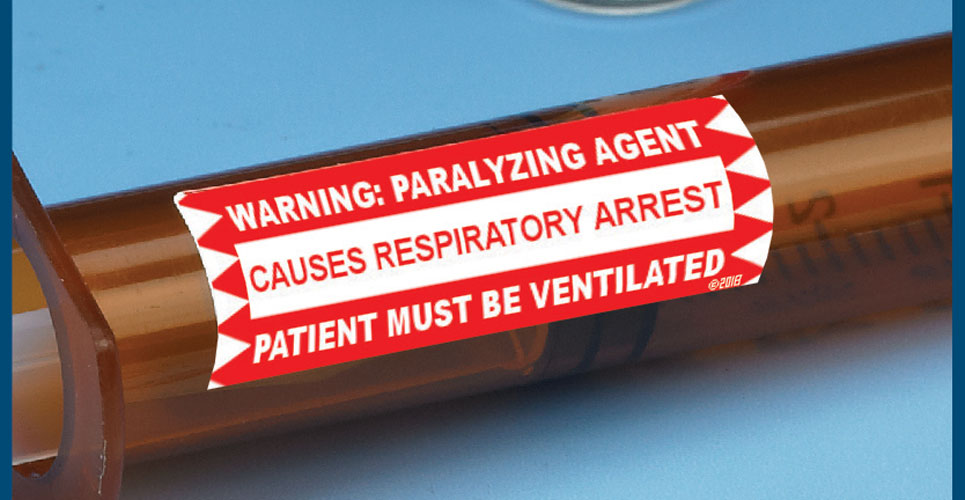
Neuromuscular blocking agents have a well-documented history of causing catastrophic injuries or death when inadvertently administered to adult and paediatric patients who were not receiving proper ventilatory assistance.
Because neuromuscular blockers paralyse the muscles necessary for breathing, some patients have died or sustained serious, permanent injuries if the paralysis was not witnessed by a practitioner who could intervene.
To help pharmacists comply with the recommendations of ISMP and other authorities, Medi-Dose has created a series of Neuromuscular Blocking Agent Warning Labels available in preprinted sheets. By recommendation, auxiliary labels stating “WARNING: PARALYZING AGENT – CAUSES RESPIRATORY ARREST – PATIENT MUST BE VENTILATED” should be placed on all storage bins and final medication containers (for example, vials, syringes, IV bags) of neuromuscular blocking agents to clearly communicate that respiratory paralysis will occur and ventilation is required.
Additionally, the same images are available for customers who use our MILT software at www.MediDose.com/NBAW so they can easily add the warning to their unit dose packaging and labelling.
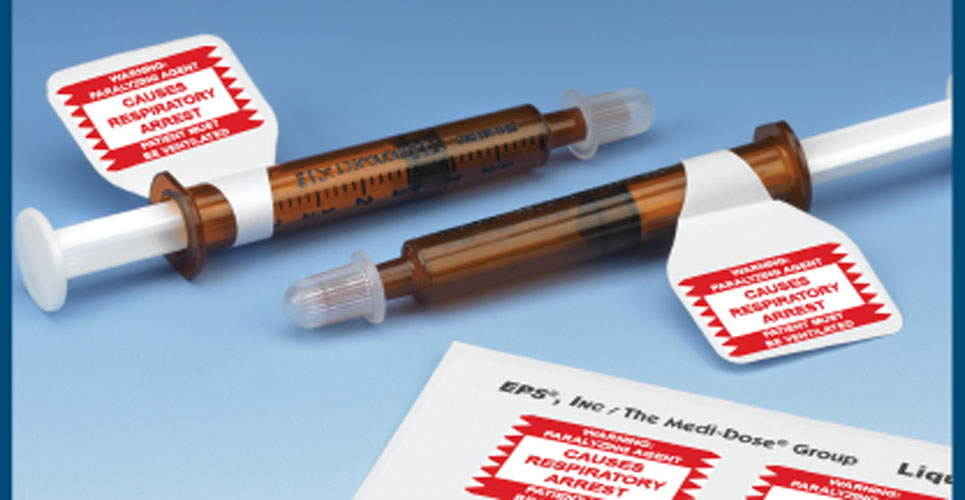
“Often the most effective means of communication between health care professionals is through labelling”, notes Robert Braverman, President of Medi-Dose. “Our Neuromuscular Blocking Agents Warning Labels were created to help health care practitioners alert one another and their patients to these new concerns when this medication is dispensed. Like our existing line of auxiliary warning labels and images, this series of labels help call attention to specific medication information to promote safety, increase awareness and reduce errors.”
For more information on the Medi-Dose Neuromuscular Blocking Agent Warning Labels and the MILT software, please contact Robert Braverman or visit www.medidose.com.