teaser
Metronic Inc. last Friday announced that it has received approval from the U.S. Food and Drug Administration (FDA) for the Endeavor® Zotarolimus-Eluting Coronary Stent System to be used in the treatment of coronary artery disease.
The approval was based on a comprehensive body of scientific evidence and gives US cardiologists access to a new medical device for safely and effectively treating patients with narrowed coronary arteries.
The Endeavor data encompasses the largest, most wide-ranging patient population submitted to the FDA in support of a drug-eluting stent, including more than 4,100 patients, followed up for as long as four years.
This extensive clinical research has shown that Endeavor provides a consistent and sustained reduction in the need for repeat procedures compared to a bare-metal stent, while also maintaining an excellent safety profile. The Endeavor stent is the first new drug-eluting stent approved by the FDA since 2004.
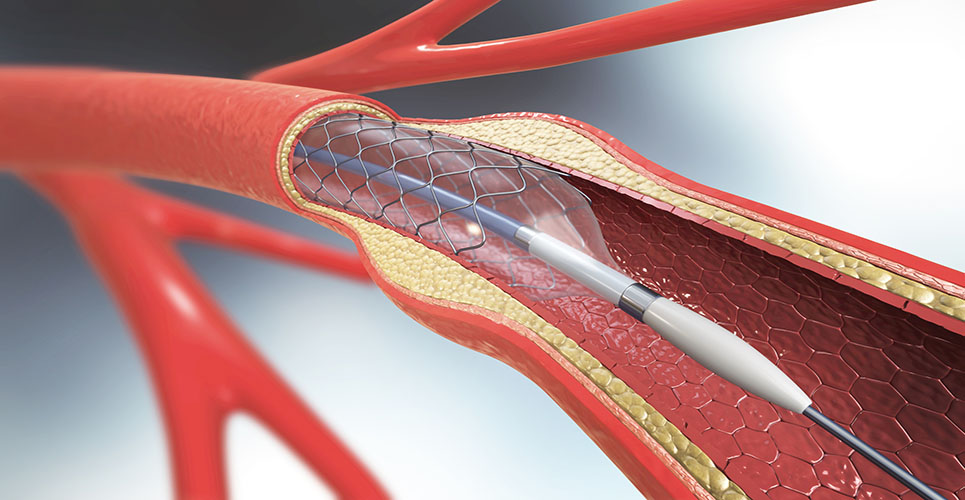
Enabling a minimally-invasive alternative to open-heart bypass surgery, stents are tiny wire mesh tubes used to prop open coronary arteries that have been cleared of blockages to restore blood flow to the heart muscle.
Drug-eluting stents deliver medication to the artery wall to reduce the chance that the artery will narrow again and require a repeat procedure.
“Endeavor represents a true next-generation drug-eluting stent because of its unique design, which combines an advanced stent platform with the potent drug zotarolimus and a biocompatible polymer,” said Dr Martin B. Leon, professor of medicine at Columbia University Medical Center in New York, cardiologist at New York-Presbyterian Hospital/Columbia University Medical Center, founder and chairman emeritus of the Cardiovascular Research Foundation, and principal investigator of the ENDEAVOR III and IV clinical trials.
“This device addresses an important need by demonstrating comparable clinical effectiveness to a first generation drug-eluting stent while also exhibiting a safety profile more typical of a bare-metal stent, long considered a benchmark for safety performance.
The S market launch of the Endeavor stent begins immediately. “We have been strengthening our field and manufacturing capabilities in anticipation of considerable demand for the Endeavor stent in the United States,” said Scott Ward, president of the CardioVascular business at Medtronic.
“We expect to ship 100,000 units to US hospitals in the next 30 days to assure full availability of this next-generation technology.”
Take part in our prize-draw survey