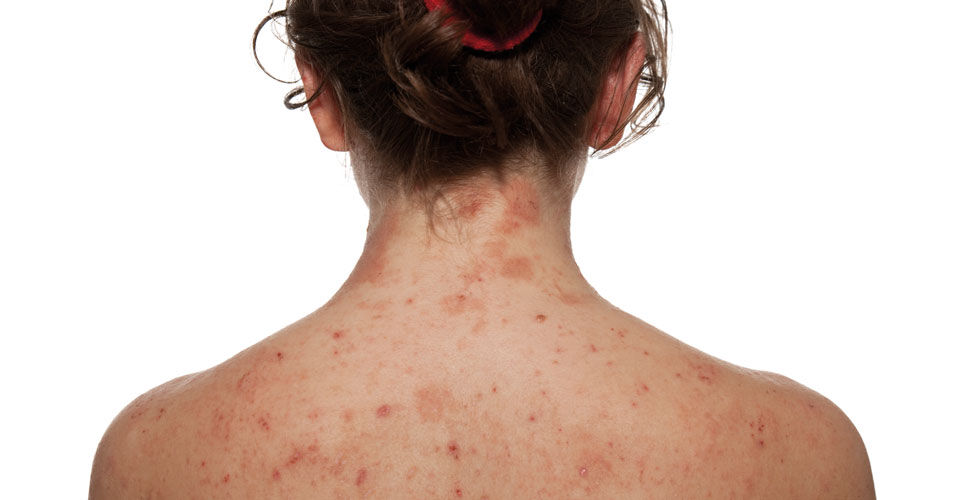
Here we report on a retrospective study undertaken in a UK hospital to investigate adherence to National Institute for Health and Care Excellence (NICE) criteria and guidance in treatment of atopic dermatitis with a biologic therapy
Atopic dermatitis (AD) is a chronic, recurrently flaring, generalised skin condition that can be life-limiting, debilitating and isolating. It can affect all aspects of life (physical, psychological, social and financial). Severe disease is associated with intolerable itch that disrupts sleep, and there is a higher risk of depression and suicide.1 It affects 2%–10% of adults worldwide and can be associated with other systemic disorders such as asthma.2
The severity of atopic dermatitis and its effect on quality of life is assessed by a variety of scoring systems that include both subjective and objective measurements.3 A typical treatment pathway involves emollients and topical corticosteroids (first line), topical calcineurin inhibitors (i.e., tacrolimus, pimecrolimus) as second line, phototherapy (third line) and systemic immunosuppressant therapies (fourth line) including oral corticosteroids, ciclosporin (the only licensed drug), methotrexate, azathioprine and mycophenolate mofetil. Long term use of systemic agents is not recommended due to the risk of adverse side effects.4
Dupilumab was the first therapy to be approved for moderate-to-severe AD that does not respond to topical therapies based on large, randomised, double-blind placebo-controlled clinical trials.5–7 Dupilumab (Dupixent, Sanofi Genzyme) is a human monoclonal antibody that targets the signalling mechanisms of cytokines interleukin (IL)-4 and IL-13. It is indicated for the ‘treatment of moderate to severe atopic dermatitis in adults who are candidates for systemic therapy’. It is given by subcutaneous injection and the recommended dose is 300mg every two weeks after an initial loading dose of 600 mg (two 300mg injections at different sites). Various national guidelines position dupilumab either after other systemic therapies6 or as another systemic therapy option in the pathway for uncontrolled atopic dermatitis.7–9
Across England and Wales, guidance from the National Institute for Health and Care Excellence (NICE) assesses the clinical and cost effectiveness of health technologies, including pharmaceuticals. The National Health Service (NHS) is legally obliged to fund and resource medicines and treatments recommended by NICE’s technology appraisals (TA). Furthermore, the NHS Constitution, which sets out rights to which patients, public and staff are entitled, and pledges which the NHS is committed to achieve, states that patients have the right to drugs and treatments that have been recommended by NICE for use in the NHS, if their doctor believes they are clinically appropriate.8
Within the NICE TA, dupilumab is recommended as an option for treating moderate to severe AD in adults, only if: the disease has not responded to at least one other systemic therapy, such as ciclosporin, methotrexate, azathioprine and mycophenolate mofetil, or these are contraindicated or not tolerated; and the company provides dupilumab according to the commercial arrangement. Dupilumab should be stopped at 16 weeks if the AD has not responded adequately. An adequate response is: at least a 50% reduction in the Eczema Area and Severity Index (EASI) score from when treatment started; and at least a 4-point reduction in the Dermatology Life Quality Index (DLQI) from when treatment started.1
EASI assesses both lesional intensity and extent. Four signs – erythema, papulation/oedema, excoriation, and lichenification – are assessed using a scale of 0 to 3. EASI assesses the average lesional intensity within four body regions:
- head and neck
- arms
- trunk
- legs.
Lesional extent is evaluated by estimating the surface area involved of those four body regions (1%–9%, 10%–29%, 30%–49%, 50%–69%, 70%–89%, and 90%–100%). Lesional intensity is multiplied by the surface area involved in that region and summed across regions, yielding a total score ranging from
0 to 72.9 DLQI consists of 10 queries that assess the patient’s perception of the effects of the specific skin condition on their health-related quality of life over the past week. Summing the score of each question resulting in a maximum of 30 and a minimum of 0. The higher the score, the more quality of life is impaired.10
The NHS price for dupilumab excluding VAT is £1,264.89 for 2x300mg, although a patient access scheme is in place as part of the NICE TA. A patient access scheme or commercial arrangement associated with the NICE guidance is a way for pharmaceutical companies to lower the acquisition cost to the NHS to improve its cost-effectiveness, so enabling patients to gain access to high-cost medicine treatments. High-cost medicines are so named as they are expensive prescribed items, representing a disproportionate cost relative to the total NHS cost of the relevant hospital episode in terms of volume and cost.
The hospital
Royal Cornwall Hospitals NHS Trust is a 750-bed acute teaching district general hospital in the south-west of England. The hospital, in conjunction with the local clinical commissioning group (CCG, now integrated care board), utilises the Blueteq high-cost drug management system. CCGs were statutory regional NHS bodies responsible for the planning and commissioning of healthcare services for their local area. Many CCGs in England use this Blueteq web-based system which allows clinicians to complete an online proforma for patients prescribed a high-cost medicine and receive automatic approval for funding if the patient meets all the relevant criteria which normally reflect the NICE TA guidance. This ensure that clinicians receive the approval to treat immediately. The Blueteq system retains, as an audit trail, the request history, including patient name, drug, indication, criteria for use, date of request, requesting clinician, and whether the request was granted or not. This enables commissioners to monitor the use of expensive treatment, so that only treatments prescribed in line with NICE guidelines are reimbursed to the hospital.
Aim
We aimed to identify whether dupilumab had been used according to the criteria based on the NICE TA.
Methods
This was a retrospective, single site study in an acute teaching district general hospital. An extract was downloaded from the Blueteq system for adult patients granted approval to commence dupilumab treatment for moderate to severe atopic dermatitis, and who were then potentially eligible to receive such treatment for more than 16 weeks. Relevant data (patient demographics and treatment details) were imported into Excel by a member of the pharmacy team. Dermatology correspondence (as part of the medical record) were examined as far back as 2012 if necessary for relevant information about patients’ treatment.
Health Research Authority criteria about research and service evaluation were considered. This was a retrospective assessment involving no changes to the service delivered to patients, and we used the NHS Health research authority tool (www.hra-decisiontools.org.uk/research/index.html) which helped confirm that no ethical approval was required for this project. Patient data were used in accordance with local NHS hospital policy.
Results
At the time of the Blueteq extract, there were 88 patients on the Blueteq system, and the records of 30 patients were reviewed. Most entries were from when the NICE TA first approved dupilumab, though a few entries were chosen from late 2021/early 2022
There were 18 males and 12 females who had commenced on dupilumab and been reviewed approximately 16 weeks later; mean age was 44 years (range 18–73).
Twenty-six (87%) patients had tried phototherapy and 26 (87%) had evidence in their dermatology records of having been prescribed a course of prednisolone at some point. As regards the named prior systemic therapy (ciclosporin, methotrexate, azathioprine, and mycophenolate) in the NICE TA, all but one patient had been on at least one of these medicines. Four patients had tried all four therapies.
One patient, who began dupilumab at a tertiary centre, did not have a starting EASI score in our hospital records. Of the 29 patients with a baseline score, 28 (97%) experienced the required 50% or more reduction in EASI score at the review meeting, whilst 28 (93%) of all 30 patients achieved the at least 4-point reduction in DLQI. One patient did not meet both NICE criteria showing only a reduction in EASI score from 33.6 to 27.9 (and not the required 50% reduction) and DLQI from 30 to 28 (and not the 4-point reduction) , and one patient had the required reduction in EASI score from 48 to 2 but did not have the required DLQI reduction (only a decrease from 13 to 10 and not the 4-point reduction ). Both had their treatment ceased. Table 1 shows overall results at baseline and at review.
Discussion
This small-scale study found that 28 of 30 patients met all the NICE starting requirements for dupilumab treatment. The two instances of non-compliance were one patient who did not have recorded locally an EASI score, and one patient who did not try any prior systemic therapy. This latter patient, whilst waiting for an appointment to commence ciclosporin, was admitted acutely to hospital with severe atopic dermatitis and started on dupilumab as this was deemed to be quicker acting with no need to wait for the necessary blood tests associated with immunosuppressive therapy.
Two patients who did not meet the reduction in EASI or in DLQI at review both had their treatment ceased. The drug cost impact of continuing inappropriate treatment in these two patients would be about £33,000 per year at NHS price and excluding VAT as these are provided via homecare. The actual cost impact is less than this due to the commercial arrangement in place.
A similar UK based study reported on 30 patients who received dupilumab after one or more immunosuppressive drugs.11 Their patients had average scores of 30 on EASI and 15 on the DLQI at baseline (compared to our study values of 29.7 and 18.9, respectively). All their 30 patients showed adequate response at 16 weeks and therefore continued to receive dupilumab. They report a mean 70% reduction in EASI after 16 weeks, and a 4-point or more reduction in DLQI for all the 30 patients. Of our 29 patients with a baseline EASI score, we saw at least a 70% reduction in 26 patients, with a further 2 patients achieving the NICE requirement of at least a 50% reduction and the one patient who did not achieve this reduction had therapy stopped. Two of our 30 patients did not meet the required DLQI score reduction. The patient who did not achieve the DLQI reduction had severe side effects from dupilumab and this would have adversely influenced the DLQI score. This patient with a poor DLQI response moved onto baricitinib as another option approved by NICE.12
The reported results from our small study are not directly comparable with the major controlled trials for dupilumab which enrolled different groups of patients. SOLO1 and SOLO2 enrolled adults with moderate-to-severe atopic dermatitis whose disease was inadequately controlled by topical treatment.5 LIBERTY AD CHRONOS included patients with moderate-to-severe atopic dermatitis and an inadequate response to topical corticosteroids. These patients were treated with topical corticosteroids as well as dupilumab or placebo.6 LIBERTY AD CAFÉ included patients with a history of inadequate response or intolerance to ciclosporin, or for whom ciclosporin treatment was medically inadvisable.7 For these trials, the Dupixent Summary of Product Characteristics reports on mean EASI score reductions at week 16 of approximately 72%, 67%, 80% and 80%, respectively.13 Overall, we saw a 90% reduction in EASI score for the 29 patients with both a baseline and review score.
Other real-world evidence has looked at efficacy and safety at 52 weeks.14,15 These reports may have investigated different cohorts of patients and different efficacy measures, making direct comparisons difficult. Recognising that long-term ongoing treatment in patients with persistently controlled atopic dermatitis might lead to overtreatment and an increase in adverse events (e.g., injection site reactions, conjunctivitis), there is some limited research into how tapering of dupilumab dosing should be undertaken.16
As regards use of the EASI score: because this has moderate inter-observer reliability, it was suggested that the same investigator should perform the EASI in a patient at baseline and follow-up.17 This does not always occur in our centre; however, use of a tool such as EASI score.com18 (which has picture guidance) helps to reduce the subjectiveness of the scoring process.
We recognise the limitations of a single centre, very small-scale retrospective study that sampled approximately 34% of the patients recorded on Blueteq as having commenced dupilumab. We do not report prolonged follow up of patients in relation to any improvement in their atopic dermatitis other than what we observed documented in dermatology letters at commencement and review of treatment. These results therefore cannot be generalised to other hospitals.
Conclusion
In this very small study, 28 of 30 patients were compliant with NICE criteria for commencing dupilumab in atopic dermatitis. Two patients who did not achieve the required improvement at the 16-week review and had their treatment ceased.
Authors
Michael Wilcock BSc (Hons) MPhil
Leanne Roberson MAPharmT
Alison McInnes DIP(HE)Nursing/NIP
Pharmacy and Dermatology Departments, Royal Cornwall Hospitals NHS Trust, Truro, UK
Declaration of interests: The authors have no interests to declare
Key points
- Dupilumab is indicated for the treatment of moderate to severe atopic dermatitis in adults who are candidates for systemic therapy.
- National Institute for Health and Care Excellence (NICE) technology appraisal guidance describes starting and stopping criteria in adults.
- In this retrospective study of dermatology correspondence, we found 28 of 30 patients were compliant with NICE criteria for commencing dupilumab in atopic dermatitis.
- Two patients who did not achieve the required improvement at the 16-week review and had their treatment ceased.
- Seeking assurance that this payment-by-results-excluded drug is used according to NICE guidance is important for the commissioners as well as internally for the hospital and the department.
References
- National Institute for Health and Care Excellence. Dupilumab for treating moderate to severe atopic dermatitis. TA534, August 2018. www.nice.org.uk/guidance/ta534 (accessed November 2022).
- Silverberg JI, Hanifin JM. Adult eczema prevalence and associations with asthma and other health and demographic factors: a United States population-based study. J Allergy Clin Immunol 2013;132:1132–8.
- Ud Din AT et al. Dupilumab for atopic dermatitis: the silver bullet we have been searching for? Cureus. 2020;12(4):e7565.
- Sidbury R et al. Guidelines of care for the management of atopic dermatitis: section 3. Management and treatment with phototherapy and systemic agents. J Am Acad Dermatol 2014;71:327–49.
- Simpson EL et al. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med 2016;375:2335–48.
- Kulthanan K et al. Clinical practice guidelines for the diagnosis and management of atopic dermatitis. Asian Pac J Allergy Immunol 2021;39:145-–55.
- Wollenberg A et al. ETFAD/EADV Eczema task force 2020 position paper on diagnosis and treatment of atopic dermatitis in adults and children. J Eur Acad Dermatol Venereol 2020;34:2717–44.
- National Institute for Health and Care Excellence. NICE technology appraisal guidance. www.nice.org.uk/About/What-we-do/Our-Programmes/NICE-guidance/NICE-technology-appraisal-guidance (accessed November 2022).
- Chopra R, Silverberg JI. Assessing the severity of atopic dermatitis in clinical trials and practice. Clin Dermatol 2018;36:606–15.
- Dermatology Life Quality Index. www.cardiff.ac.uk/medicine/resources/quality-of-life-questionnaires/dermatology-life-quality-index (accessed November 2022).
- Deif E, Bali S, Rajeev A. Dupilumab in the treatment of moderate-to-severe atopic dermatitis: A focused review. J Skin Sex Transm Dis 2021;3:151–5.
- National Institute for Health and Care Excellence. Baricitinib for treating moderate to severe atopic dermatitis. TA681, March 2021. www.nice.org.uk/guidance/ta681 (accessed November 2022).
- Dupixent 300mg solution for injection in prefilled pen. www.medicines.org.uk/emc/product/11321/smpc#gref (accessed November 2022).
- Jo CE et al. Evaluation of long-term efficacy, safety, and reasons for discontinuation of dupilumab for moderate to severe atopic dermatitis in clinical practice: A retrospective cohort study. J Am Acad Dermatol 2020;82:1530–2.
- Napolitano M et al. Efficacy and safety of dupilumab in clinical practice: one year of experience on 165 adult patients from a tertiary referral centre. Dermatol Ther (Heidelb) 2021;11:355–61.
- Spekhorst LS et al. Patient-centered dupilumab dosing regimen leads to successful dose reduction in persistently controlled atopic dermatitis. Allergy 2022; Jul 15: doi: 10.1111/all.15439 (online ahead of print).
- Schmitt J et al. The Harmonising Outcome Measures for Eczema (HOME) statement to assess clinical signs of atopic eczema in trials. J Allergy Clin Immunol 2014;134:800–7.
- www.EasiScore.com/ (accessed November 2022).