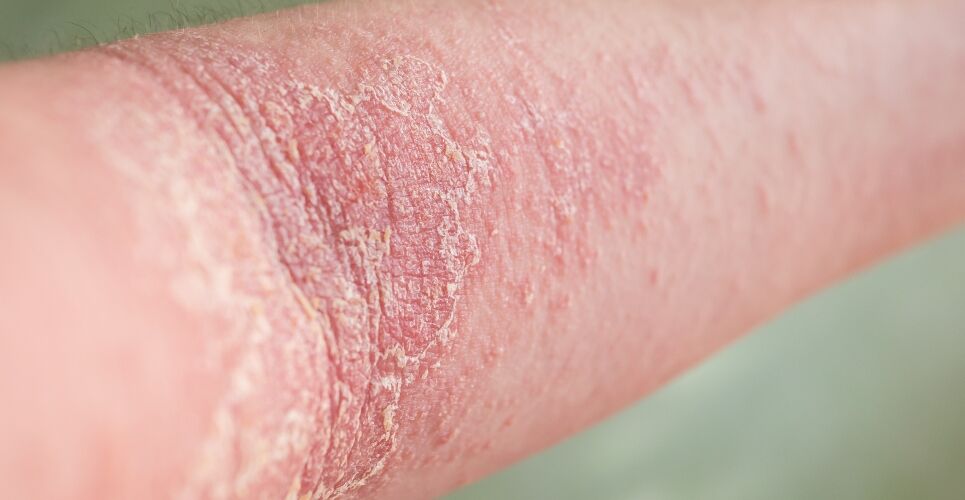
Amlitelimab has met its primary endpoint of an improvement in disease severity of atopic dermatitis in a recent phase 2b dose-ranging trial, its manufacturer Sanofi has announced.
Amlitelimab provided statistically significant improvements in signs and symptoms of moderate to severe atopic dermatitis in adults whose disease could not be adequately controlled with topical medications or where topical medications are not a recommended treatment approach.
A non-depleting IgG4 human anti-OX40L monoclonal antibody, amlitelimab’s OX40–OX40L interaction appears to play a central role in the pathogenesis of atopic dermatitis. The drug binds to OX40L and in doing so, blocks its interactions with OX40, thus restoring immune homeostasis between pro-inflammatory and anti-inflammatory T-cells. Consequently, the drug offers a potential first-in-class novel antibody therapy for patients with moderate to severe atopic dermatitis.
Amlitelimab and atopic dermatitis disease severity
Amlitelimab (formerly KY1005) was found in a pharmacokinetic study to reduce skin redness, indicating its potential as a novel pharmacological treatment in immune-mediated disorders. Earlier work revealed how amlitelimab leads to a significant reduction of interleukin-13 levels with a corresponding reduction in dermatitis disease severity. Furthermore, the drug also improves disease severity through targeting interleukin-22.

The latest information released by Sanofi relates to STEAM-AD – a phase 2b, double blind, five-arm study assessing amlitelimab in adult participants with moderate to severe atopic dermatitis. While the company did not provide specific details, it did report that amlitelimab had enrolled 390 people from several countries. The primary endpoint was the percentage change in the Eczema Area and Severity Index score from baseline at 16 weeks.
The detailed efficacy and safety results from the trial are to be presented in a future scientific forum and, as amlitelimab is currently under clinical investigation, the safety and efficacy have not yet been evaluated by regulatory authorities.
Dr Naimish Patel, head of global development, immunology and inflammation at Sanofi, said: ‘While we have made significant strides in the treatment of atopic dermatitis, there are patients who are still in need of new options.
‘We believe that the results from this Phase 2b study with amlitelimab support our perspective that targeting OX40-Ligand has the potential to provide a first and best-in-class treatment option that addresses type 2 and non-type 2 inflammation to meet the individual needs of people living with atopic dermatitis and other chronic inflammatory diseases.
‘We look forward to advancing into a larger Phase 3 clinical development programme and continuing to drive momentum in our immunology pipeline to deliver first or best-in-class treatments.’